Can Blockchain Improve Drug Safety in Supply Chains?

You purchase medicine from a pharmacy with sealed packaging, proper branding, and even a traceable batch code, and yet the treatment fails within days because the product turned out to be counterfeit.
This is the reality behind fake medicines: appearance and documentation can be convincing while the product itself is unsafe. As supply chains become more digital, the expectation is that such failures should reduce.
However, the problem persists, raising a key question: why does increased digitisation not fully prevent counterfeit drugs?
The real weakness in medicine supply chains
Medicines often move through long distribution networks involving manufacturers, logistics companies, wholesalers, retailers, and healthcare facilities.
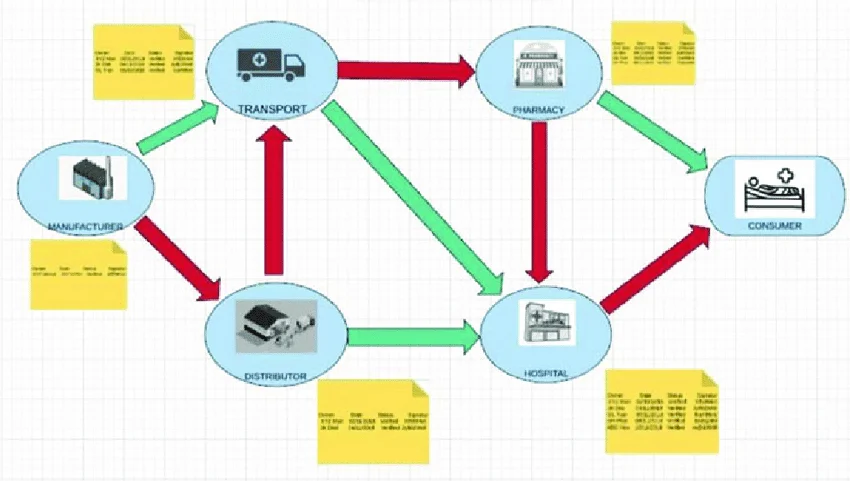
Each stage introduces a point where errors or manipulation can occur. In many regions, these processes are not fully integrated.
Some records are digital, others are paper-based, and many systems do not communicate with each other. This fragmentation makes it difficult to track products consistently from production to the patient.
Counterfeiters exploit these gaps by inserting fake products into legitimate distribution routes. Once inside, these products often move alongside genuine stock without immediate detection.
How blockchain is intended to work
Blockchain is a shared digital ledger where data is recorded across multiple systems in a way that makes later alteration extremely difficult.
In pharmaceutical supply chains, it is designed to create a continuous, verifiable history of each medicine batch.
A typical model would work like this:
A manufacturer registers a batch with a unique identifier
Each transfer between distributors and warehouses is recorded
Pharmacies log receipts before dispensing
The end user verifies authenticity using a code linked to the ledger
This creates a transparent chain of custody. If a batch is flagged, its movement history can be reviewed quickly, reducing the time needed for investigation or recall.
The system’s value lies in coordination, with multiple participants accessing the same version of transactional history instead of isolated records.
Where blockchain falls short

Despite its structure, blockchain does not verify the physical authenticity of medicines. It only secures the accuracy of recorded entries.
If counterfeit drugs are entered into the system as genuine at the point of registration or early distribution, blockchain will preserve that record without detecting the deception.
The system ensures consistency of data, not the truthfulness of inputs.
This creates a critical limitation: the technology cannot correct false information introduced at the source. Another constraint is adoption.
For blockchain to function effectively, every participant in the supply chain must consistently use it. In fragmented or under-resourced environments, partial adoption weakens the system and creates parallel channels that bypass verification.
There is also the issue of infrastructure. Reliable connectivity, standardised systems, and trained operators are required at every stage. Without these, implementation becomes inconsistent and less effective.
What still determines drug safety

Blockchain can strengthen traceability, improving how pharmaceutical supply chains are recorded and monitored, particularly in tracking movement and reducing hidden gaps, but it does not replace core regulatory and physical controls.
Drug safety still depends on:
Regulatory enforcement and inspections
Laboratory testing of products in circulation
secure packaging and serialization systems
coordinated oversight by agencies such as the National Agency for Food and Drug Administration and Control in Nigeria
consumer-level verification tools such as authentication codes or QR systems
These mechanisms address both the physical and administrative dimensions of the problem. Without them, digital tracking systems remain incomplete.
You may also like...
Can Blockchain Improve Drug Safety in Supply Chains?

Blockchain is being used to improve transparency in pharmaceutical supply chains, but questions remain about its ability...
10 Startups Just Secured $560,000 to Combat The Heat Crisis In Nigeria

Ten Nigerian startups secure $560,000 to develop solutions tackling extreme heat across agriculture, healthcare, and inf...
Sports Stars Dazzle at Met Gala 2026: Venus Williams and Alysa Liu Lead Fashion Charge!

The 2026 Met Gala celebrated "Costume Art" with a strong showing of athletes, from co-chairs like Venus Williams to fash...
Premier League Shocker: Everton Draw Puts Man City Title Race in Jeopardy!

The Premier League title race took a dramatic turn after Manchester City's 3-3 draw with Everton, significantly boosting...
Christopher Nolan's 'The Odyssey': New Trailer Unleashes Mythical Footage, Director Calls Homer 'Original Marvel!'

Christopher Nolan's highly anticipated 13th feature, "The Odyssey," has unveiled a new trailer, showcasing its epic scop...
Met Gala 2026: Unforgettable Fashion, Bold Statements & Unexpected Protests Rock the Red Carpet!

The 2026 Met Gala, themed "Fashion is Art," showcased opulent celebrity fashion amidst controversy surrounding lead spon...
Beyond the Mic: Uganda's Bobi Wine Reveals Literary and Lyrical Influences Shaping His Political Power

Ugandan music star and political leader Bobi Wine, currently in exile, discusses his 'people power' movement and the int...
Seun Kuti Declares Lifelong Feud with Wizkid Over Fela Legacy Clash

Afrobeat singer Seun Kuti has declared his rift with music star Wizkid to be permanent, stating that reconciliation is i...





