4 Documented Ozempic Dangers That Should Scare You More Than Your Weight
They called it a miracle. Celebrities posted their before-and-afters. Pharmacies ran dry. People ordered unregulated versions online and jabbed needles into their stomachs in bathroom mirrors, chasing a body they had seen on a red carpet.
Between 2022 and 2024, Ozempic became the most talked-about drug on earth, not because of the disease it was designed to treat, but because of the body it promised to sculpt.
What nobody talked about nearly enough was what that body would cost.
Here are seven documented dangers of Ozempic that deserve far more attention than they are getting.
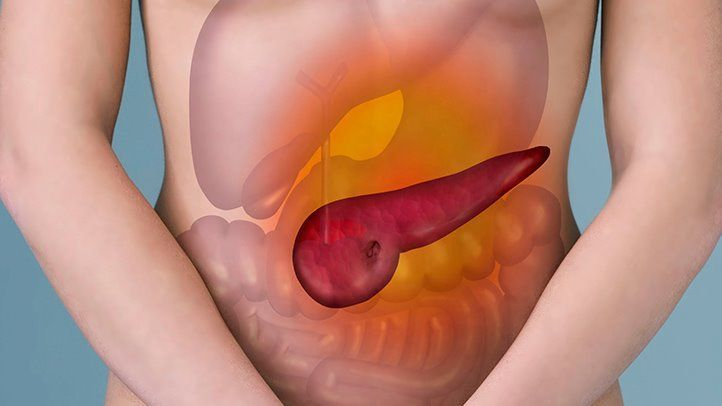
1. Gastroparesis (Stomach Paralysis)
The most brutal side effect of Ozempic is not the nausea everyone jokes about. It is gastroparesis, a condition in which the stomach essentially stops emptying. Food sits inside it, fermenting, refusing to move.
The pain is severe enough to send people to emergency rooms convinced they have appendicitis.

The FDA added a warning about intestinal obstruction to the drug's label after receiving reports of at least twenty cases of ileus, a potentially fatal bowel blockage, and at least two connected deaths. For some patients, the condition persists long after they stop the drug entirely.
2. Acute Kidney Injury
Ozempic's intense gastrointestinal side effects, vomiting, diarrhea, and loss of appetite, can cause severe dehydration. The kidneys, which depend on adequate fluid to function, bear that burden directly.
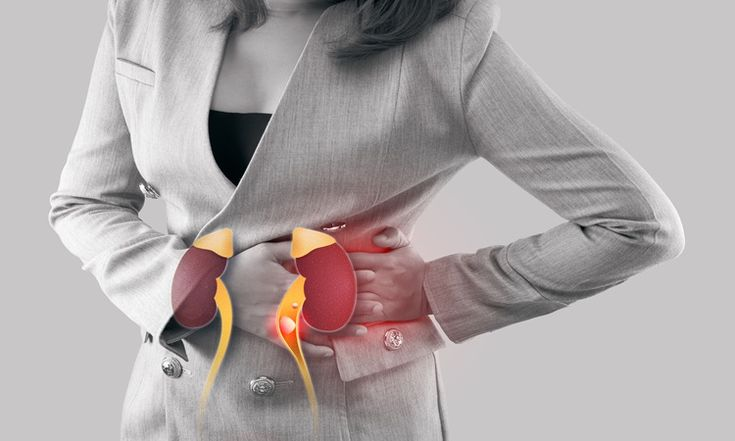
Acute kidney injury has been documented in Ozempic users, and while many recover with treatment, some cases have caused permanent damage.
3. Thyroid Cancer
Ozempic carries a boxed warning, the FDA's most serious category of safety alert, about thyroid cancer. Animal studies showed semaglutide caused thyroid tumors in rodents.
Whether that risk translates to humans is still unknown because the long-term studies at scale have not been completed. Patients are instructed to report any lump in the neck, hoarseness, or difficulty swallowing immediately.
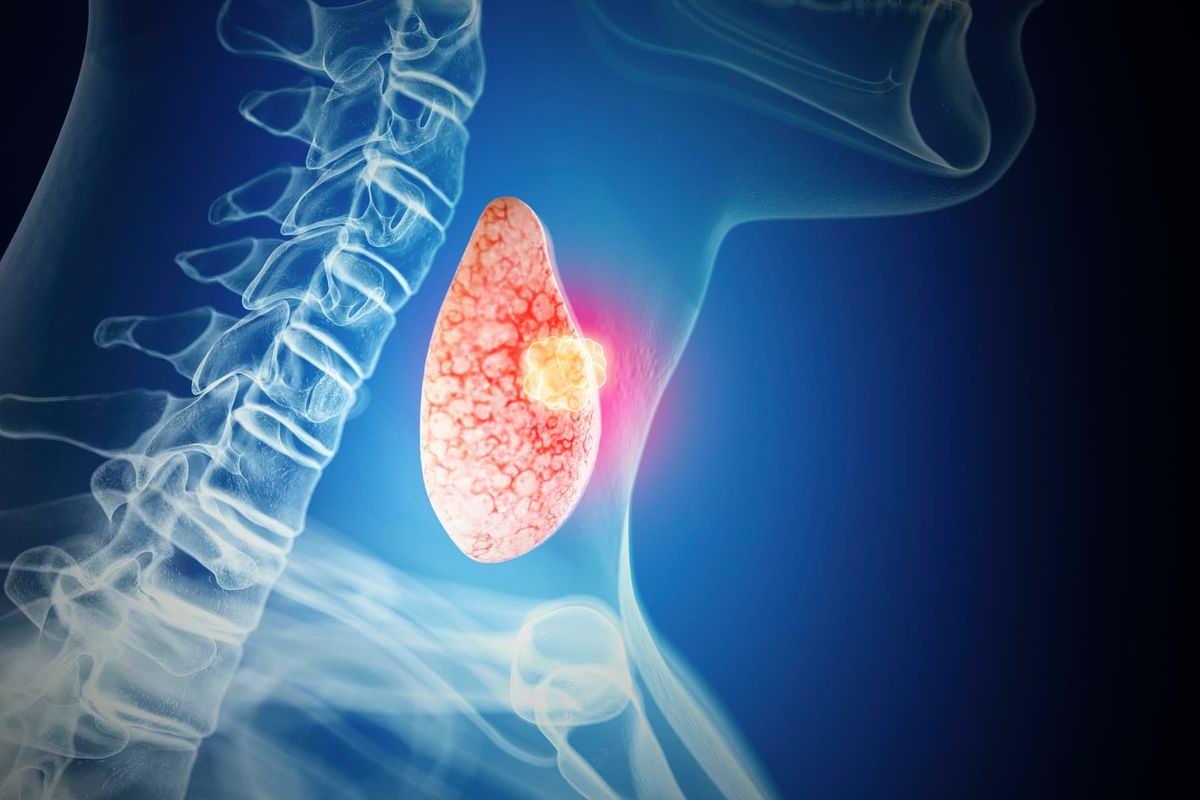
The science is not settled. For a drug being taken by millions of people for weight loss, that is a deeply uncomfortable place to be.
4. Pancreatitis
Inflammation of the pancreas has been reported in patients on semaglutide since the drug's early clinical trials. Severe upper abdominal pain radiating to the back is the warning sign, and in serious cases it has led to hospitalization.
The connection to Ozempic remains disputed in some studies, but the FDA still requires the risk to be disclosed on the label. When a drug's own manufacturer is required to warn about pancreatitis, it deserves more than a footnote in a telehealth consultation.
Conclusion
The needle is small. The consequences are not. Ozempic was designed for people with Type 2 diabetes, not for everyone chasing a number on a scale.
The drug is not inherently evil, but the culture surrounding it has moved years ahead of the science, and the people paying the price are the ones who were never given the full story before they rolled up their sleeve.
Ask your doctor. Read the label. Know what you are signing up for.
You may also like...
Record Viewership: Clark vs. Bueckers Clash Draws Millions on ABC!

The WNBA's opening weekend garnered significant viewership, with the clash between Caitlin Clark's Indiana and Paige Bue...
Pop Superstars Madonna, Shakira, BTS Set to Ignite FIFA World Cup 2026 Halftime Show!

Global music icons Madonna, Shakira, and BTS are set to co-headline the first-ever halftime show at the FIFA World Cup 2...
Asghar Farhadi & Isabelle Huppert Captivate Cannes with 'Parallel Tales' Triumph!

Isabelle Huppert stars in Asghar Farhadi’s new French-language drama “Parallel Tales,” which premiered at Cannes to a st...
New Star Wars Film 'Mandalorian and Grogu' Stirs Buzz with LA Shoot & Divisive First Reactions!

The Star Wars franchise returns to cinemas with "The Mandalorian and Grogu," a continuation of the beloved Disney+ serie...
Global Rhythms Ignite: Shakira & Burna Boy Drop Official 2026 FIFA World Cup Anthem 'Dai Dai'!

Shakira and Burna Boy have released "Dai Dai," the official song for the 2026 FIFA World Cup, now available on all major...
2026 AMVCAs: Fashion, Stars & Unforgettable Moments You Missed!

The 12th Africa Magic Viewers’ Choice Awards redefined cultural celebration, elevating fashion into a profound expressio...
Major Shift: Nigeria Eyes Reciprocal Visa-Free Travel with Rwanda!

President Bola Tinubu and Rwandan President Paul Kagame met in Kigali to strengthen bilateral ties, with discussions foc...
Astonishing Claim: Chad's Status as a Nation Questioned!

Chad faces an acute crisis of state survival, moving beyond mere authoritarianism to a profound institutional collapse w...





