BMC Geriatrics volume 25, Article number: 88 (2025) Cite this article
Postoperative frailty is an important determinant of postoperative recovery and survival outcomes. Predicting the onset of postoperative frailty is significant importance for the rehabilitation of the elderly people after surgery. Our study aims to develop and evaluate a predictive model for postoperative frailty on the 30th day in elderly patients.
Data from seven Guangzhou hospitals were collected, encompassing 2,290 patients for analysis. This study constructed the model using LASSO regression and stepwise regression, and the optimal predictive model was selected based on comparison. Model performance was assessed through calibration curves, the area under the ROC curve (AUC), and decision curve analysis (DCA).
The final model included the following variables: American Society of Anesthesiologists (ASA) grade, intraoperative blood loss, economic income, caregiver status, sedentary behavior, cognitive function, Activities of Daily Living (ADL), postoperative hemoglobin (Hb) level, and postoperative ICU admission. The model demonstrated good discrimination, with an area under the curve (AUC) of 0.7431 (95% CI = 0.7073–0.7788) in the training set and 0.7285 (95% CI = 0.6671–0.7624) in the validation set.
According to general demographic information, lifestyle habits, and surgery-related factors, a predictive model for postoperative frailty in the elderly was constructed, which has good predictive power. This model can identify high-risk populations for postoperative frailty and provides a reference for the early detection and intervention of frailty in the elderly in clinical practice.
This study was registered on May 17, 2023, at the Chinese Clinical Trial Registry (registration number: ChiCTR2300071535).
Aging is an irreversible trend, with 13% of the population expected to be over 65 by 2050 [1], likely increasing older surgical patients [2]. Addressing the prevention and intervention of age-related conditions such as sarcopenia, frailty, and cognitive impairments to enhance the postoperative quality of life and survival outcomes in the older person will emerge as a critical topic.
Frailty is a geriatric syndrome characterized by a decline in physiological capacity across multiple organ systems, with an inability to cope with exogenous or endogenous stress [3, 4]. It was reported that the incidence of frailty in older patients could be up to 25.64% [5], with an average incidence of 13% within nonsurgical older patients [6, 7]. Frailty can cause complications, slow recovery, longer hospitalization, and higher costs [8, 9]. Considering the significant impact of frailty on the rehabilitation of the older person, the identification and timely prevention of frailty have become a research hotspot.
Most older person’s perioperative research focuses on preoperative frailty, with a lack of studies on postoperative frailty prevention, indicating gaps in the current literature. Li [10] included 406 older individuals over the age of 75 who underwent percutaneous coronary intervention (PCI), and constructed a predictive model for new-onset frailty within three days postoperatively. The final model included five variables: duration of surgery, psychological distress, frequency of daily exercise, sleep condition, and comorbidity status. But the model was constructed using a single dataset, and there was no data splitting or external validation. Liu [11] constructed a predictive model for new-onset postoperative frailty within 3 months specifically for older person with hip fractures, including 192 individuals over the age of 65. However, the results have not been subjected to validation analysis. The findings from these studies are helpful in predicting postoperative frailty in different disease. Nevertheless, the reliability and generalizability are limited by small sample sizes and simplistic validation of the results. More robust and accurate prognostic estimates for postoperative frailty are thus needed.
Given the context, we’ve assembled multicenter data to enhance sample size and representativeness, creating a clinically relevant predictive model. Our study’s goal is to construct and internally validate a nomogram predicting the risk of postoperative frailty in the older person by the 30th day, providing a straightforward tool for frailty assessment and precise targets for intervention management.
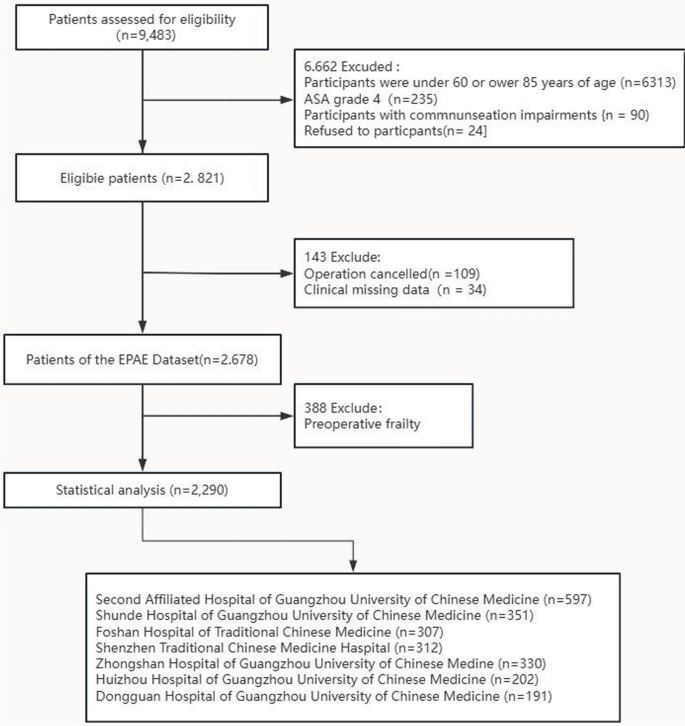
The data originate from the Early-Warning model of Perioperative Adverse Events for older adults (EPAE), which is an ongoing prospective, multicenter, longitudinal study.The project includes a total of 7 hospitals, Guangdong Provincial Hospital of Traditional Chinese Medicine, Dongguan Hospital of Traditional Chinese Medicine, Foshan Hospital of Traditional Chinese Medicine, Huizhou Hospital of Guangzhou University of Chinese Medicine, Shenzhen Traditional Chinese Medicine Haspital,Shunde Hospital of Guangzhou University of Chinese Medicine, and Zhongshan Hospital of Guangzhou University of Chinese Medine. Detailed inclusion criteria were as follows: 1) aged 60–85 years; 2) scheduled for elective surgery; 3) American Society of Anesthesiologists (ASA) classification is I–III;4) have good language expression ability, communicate with the investigators without obstacles; 5) voluntary participation in the study and signed informed consent. Exclusion criteria: 1) with severe cognitive impairment, mental dysfunctions, or communication disorders;2) cancellation of the surgery due to various reasons;3) unable to cooperate with assessment;4) Preoperative frailty population;5) Unable to cooperate while still in the ICU 30 days postoperatively. A total of 2,290 complete cases were included in the final study. For detailed procedures, please refer to the Fig. 1. All participants were randomly divided into a training set and a testing set at a ratio of 7:3. This study was registered on May 17, 2023, at the Chinese Clinical Trial Registry (registration number: ChiCTR2300071535).
The 5-item Frail scale was used to assess physical frailty [12]. The FRAIL scale is valid for the older Chinese adult population [13]. The Frail scale scores range from 0–5, with one point for each item. Patients with ≥ 3 points are deemed frail, 1–2 points are prefrail, and 0 points are non-frail.
Socio-demographic variables included age, gender, marital status, educational level, family per-capita monthly income, caregivers, smoking, drinking, dietary habit, resistance training, and sedentary behavior (a sedentary time of ≥ 6 h/day). Gender was defined as either male or female. Education level was categorized as “primary school and below”, “junior high school”, “high school or vocational training”, or “college and above” Marital status was classified as living with a spouse or without a spouse if the participant was currently separated, divorced from a spouse, widowed, or never married. Smoking and alcohol history were defined as “yes,” “quit,” or “no” based on the participant’s smoking and drinking behavior over the past year. Sedentary behavior and exercise habits were dichotomized into “yes” or “no” categories. Sedentary behavior was defined as sitting for 6 h or more per day, and exercise habit was defined as engaging in resistance training for more than 40 min per week. Eating habits were categorized into three types: predominantly vegetarian, balanced (vegetarian and non-vegetarian), and predominantly meat-based. Predominantly vegetarian refers to consuming more than 70% plant-based foods daily. Balanced (vegetarian and non-vegetarian) indicates that animal and plant-based foods are approximately equally represented in the daily diet. Predominantly meat-based refers to more than 70% of daily food intake coming from meat, dairy, and eggs [14].
Clinical and physical variables included body mass index(BMI), polypharmacy (defined as concurrent five or more drug usage) [15], history of stress(defined as experiencing at least one of the stressful events, such as severe trauma, infection, hospitalization, death of a spouse, etc.), the Age-Adjusted Charlson comorbidity index(ACCI) [16, 17], Nutritional Risk Screening 2002(NRS2002) [18], Numerical Rating Scale of pain (NRS) [19], Social support rating scale(SSRS) [20], Athens Insomnia Scale(AIS) [21], activities of daily living (ADL) [22]. Laboratory indicators include hemoglobin (Hb), Neutrophil–lymphocyte ratio (NLR) were assessed. Among these, BMI, ACCI Score, NRS2002 Score, NRS, SSRS Score, AIS Score, ADL Score, Hb (g/L) and NLR were included as continuous variable. Polypharmacy and history of stress were included as binary variables in the study.
Depression was measured on Patient Health Questionnaire-9 (PHQ-9) [23] which designed to screen for depression in primary care and other medical settings [24, 25].
Cognitive function was assessed by the brief cognitive test (Mini-cog) consisting of two components, a delayed three-word recall and a clock drawing test [26]. The Mini-Cog has been reported to have little potential for bias in terms of education or language [27].
Surgical-related variables were categorized into preoperative, intraoperative, and postoperative stages. Preoperative variables included ASA grade, preoperative fasting time (the time from the last meal to surgery), preoperative fasting fluid time (the time from the last intake of fluids to the start of surgery), and preemptive analgesia (defined as effective pain relief before surgical injury). Intraoperative variables included the surgical site (classified as limbs, abdomen, buttocks, back, head and neck, and chest), anesthesia method (defined as regional anesthesia or general anesthesia), surgery duration, intraoperative blood loss, intraoperative hypothermia (defined as any core temperature < 36 °C during surgery), and intraoperative hypotension (defined as mean arterial pressure < 60 mmHg). Postoperative variables included postoperative catheter placement, and ICU admission. Intraoperative hypothermia, intraoperative hypotension, postoperative ICU admission, and postoperative catheterization were recorded as “Yes” or “No”.
Data were collected by trained nurses through face-to-face questionnaires in the ward. For detailed information, please refer to Attachment 2. Demographic characteristics, basic clinical data (BMI, polypharmacy, history of stress, ACCI, NRS2002, SSRS, PHQ-9) were collected within 24 h after hospitalization. Other clinical and physical variables (NRS, AIS, ADL), cognitive variables (Mini-Cog) were collected on the first postoperative day, and frailty assessment were collected on day 30 after surgery. All laboratory indicators on the first postoperative day were extracted from the electronic medical records. Surgery-related variables were obtained from the electronic medical records within 24 h after surgery. We completed the assessment of postoperative frailty through telephone follow-up. All scale scores and laboratory indicators were included in the study as continuous variables.
To ensure consistency in multicenter data collection, we implemented several measures. All centers used a standardized case report form and personnel underwent uniform training and assessment for tasks including postoperative blood collection and questionnaire administration, with only those who passed participating in formal data collection. Weekly, three questionnaires from each hospital were randomly reviewed; if two-thirds were unsatisfactory, the hospital’s data for that week were re-audited to ensure quality. Upon completion, data from all centers were entered into an online management platform designed and instructed by hospital engineers.
Data presentation was tailored to variable types. Continuous variables were tested for normality and described by mean ± SD or median (IQR); categorical variables were reported as percentages. Normal continuous data were analyzed using Student’s t-test, and non-normal data with the Mann-Whitney U test. The Chi-square test assessed categorical variable differences. Lasso regression identified risk factors, with multicollinearity assessed by VIF (threshold 10), and addressed by adjustment or exclusion. Model discrimination and consistency were evaluated by the C-index and calibration curve. The data were randomly allocated in a 7:3 ratio to the training (n = 1602) and validation (n = 688) sets. All statistical analyses were performed with R software v4.2.1.
This study utilized LASSO regression and stepwise regression to develop a model. Initially, LASSO regression addresses model overfitting by constructing a penalty function that shrinks variable coefficients. This process compresses non-essential coefficients to zero while retaining important variables with coefficients greater than zero [28]. The optimal Lambda value and key variables are determined through 10-fold cross-validation [29]. Stepwise regression selects the most appropriate predictors by repeatedly adding or removing variables. In this study, we used a combination of forward selection and backward elimination, known as the forward-backward method, to progressively add and remove independent variables in order to identify the best model. This approach helps reduce model complexity and enhance predictive performance. To ensure the chosen model provides the best fit, we used the Akaike Information Criterion (AIC) to determine the optimal model and the predictors it includes. A lower AIC value indicates a better model fit and reduced risk of overfitting. These key variables are then included in a multivariate logistic regression, and significant predictive factors (p < 0.05) are integrated into the final nomogram model. Finally, to compare the predictive performance of the two models, we evaluated their classification ability using the area under the curve (AUC) and identified the optimal predictive model.
We used the AUC and calibration curves to assess the model’s discrimination ability and the degree of consistency between predicted probabilities. It is generally accepted that a model’s area under AUC greater than 0.7 is considered acceptable, indicating a model with a satisfactory level of discrimination ability [30, 31]. The nomogram was internally validated using the bootstrapping approach, and it ultimately gained further confirmation through the validation dataset.
The clinical validity of the model was evaluated using decision curves. All tests were two-tailed, and P < 0.05 was considered statistically significant. The missing data rate for all variables in this study was below 20%. To handle missing data, we used multiple imputation, creating five imputed datasets and comparing them. The results showed that the differences between the imputed datasets remained consistent, with a variation of approximately 5%. The third imputed dataset was selected as the final dataset and compared with the non-missing data in the original dataset. The comparison revealed that the relationships between the variables and postoperative frailty were consistent with those in the original data after imputation.
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee and with the Helsinki declaration. Ethical approval was obtained from the Institutional Review Board of the Second Affiliated Hospital of Guangzhou University of Chinese Medicine (protocol number BE2022-165-01).
All participants had been discharged from the ICU by the 30th postoperative day, but 19 participants had not yet been discharged. The average length of stay was 7.7 ± 5.5 days. In total 2290 old people after surgery were included in this study. There were 899 male patients (39.3%), 1391 female patients (60.7%). The average age is 69 (± 6) years old. The incidence of frailty was 12.4% (285/2290), 199(12.4%) and 86 (12.5%) comprised the training and testing sets, respectively. All participants had been discharged from the ICU by the 30th postoperative day, but 19 participants had not yet been discharged. The average length of stay was 7.7 ± 5.5 days.Compared to participants who did not develop frailty by postoperative day 30, those who developed frailty tended to be older, had a higher proportion of females (P = 0.02), lower income (P = 0.00), less daily physical activity (57.3% vs. 65.2%), and more sedentary behavior (37.7% vs. 26.8%). Additionally, frail patients had poorer cognitive function and lower social support scores (P < 0.05). In terms of surgical factors, patients who developed frailty were more likely to undergo higher-grade surgeries (P = 0.01), receive more blood transfusions during surgery (P = 0.00), and have lower postoperative hemoglobin levels (P = 0.00). In contrast, frail patients had a higher proportion of postoperative ICU admissions (9.0% vs. 2.4%). Further descriptive characteristics for all participants are listed in Tables 1 and 2. Comparisons between the training and testing sets are presented in Supplementary Table S1.
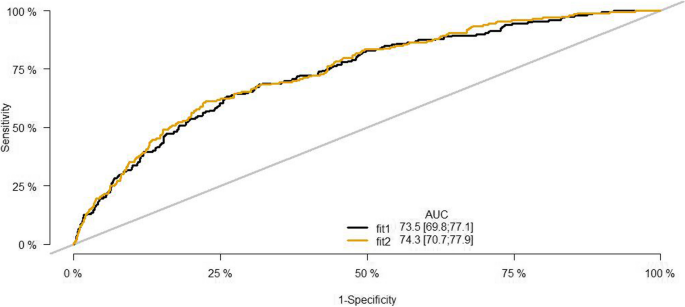
We constructed two models using LASSO regression (fit1) and stepwise regression (fit2), respectively. Through AUC comparison (Fig. 2), we ultimately selected the stepwise regression model (fit2) as the final model. The final model includes a total of 8 independent influencing factors: Mini-cog score (P = 0.02), ADL (P < 0.01), Hb(P < 0.01), average monthly household income (P < 0.01), with or without caregiver (P < 0.01), sedentary behavior (P < 0.01), ASA grade(P = 0.01), and ICU admission(P < 0.01) were associated with the development of postoperative frailty within 30 days in older person after surgery (Table 3).
The variance inflation factor (VIF) test was performed, and VIF values for all variables were < 4, indicating no covariance and the model fit was good. The predictive model was presented using a nomogram, which can be used to quantitatively predict the risk of frailty in patients after surgery (Fig. 3). The value of each of these factors was given a score on the point scale axis. A total score could be easily calculated by adding each single score and by projecting the total score onto the bottom risk scale axis, we were able to estimate the probability of frailty occurrence in the older person within 30 days post-surgery.
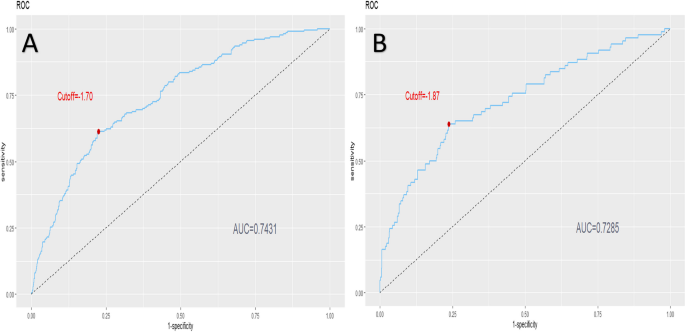
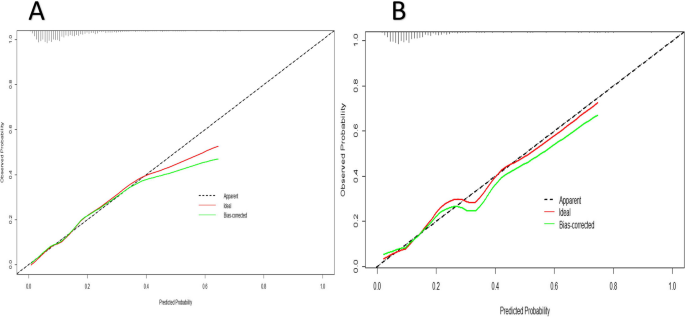
The model’s ability to discriminate postoperative frailty was evaluated using the AUC value (Fig. 4). The training set showed an AUC of 0.74 (95% CI 0.7073–0.7788), with specificity at 0.78 and sensitivity at 0.61. The validation set had an AUC of 0.73 (95% CI 0.6671–0.7755), with specificity at 0.76 and sensitivity at 0.64. For detailed results, please refer to Table 4. The Hosmer–Lemeshow test confirmed good calibration for both sets (training: χ2 = 8.157, df = 8, p = 0.4182; testing: χ2 = 5.1581, df = 8, p = 0.7406). Calibration curves are depicted in Fig. 5. The calibration curve shows that predicted probabilities of frailty closely match actual probabilities in both training and validation sets. The Decision Curve Analysis (DCA) method (Fig. 6) demonstrated that our predictive model’s net benefits surpassed those of two extreme strategies, confirming the model’s superior net benefit and predictive accuracy.
A Nomogram ROC curves generated from the training dataset. B Nomogram ROC curves generated using the testing dataset
To the best of our knowledge, this is the first study to construct a predictive model for the risk of frailty at the 30th postoperative day in older surgical patient. In this study, we have constructed a nomogram for predicting postoperative frailty using multicenter data and conducted internal validation. It includes nine predictive variables: ASA grade, with or without caregiver, Mini-cog score, ADL, sedentary behavior, ICU admission, monthly income, postoperative Hb.
Mini-cog score, ADL, Hb, monthly income, with or without caregiver, sedentary behavior, ASA grade, and postoperative ICU admission. This nomogram demonstrated good predictive performance in both the construction dataset and the validation dataset.
The occurrence of frailty in patients after surgery in the present study was 12.4%, which is significantly lower than the previously reported rate of 25.64% [5]. This may be due to our study exclude the patients with preoperative frailty or prefrail, with a focus on those who developed frailty within 30 days postoperatively. Initially, we excluded individuals with preoperative frailty, leading to a group of participants who were generally healthier and thus had a lower rate of postoperative frailty. Furthermore, unlike previous studies that concentrated solely on patients with fractures, our study encompassed a broader spectrum of surgical procedures, which accounts for the variability in the incidence rates of postoperative frailty. Additionally, we only concentrated our analysis on the emergence of frailty within the first 30 days following surgery. Frailty, as a relatively stable physical state, requires a certain period to manifest and develop [4]. Early postoperative frailty within 3 to 7 days may be due to immediate pain or temporary treatment effects and may not indicate persistent conditions. Frailty occurring later, at three or six months, might be less related to the surgery itself. Hence, we selected a 30-day postoperative period to consistently observe new-onset frailty. By this time, patients are typically past the acute recovery phase, making this window more relevant for assessing frailty development related to surgery.
Moreover, in the final predictive model, surgical factors are significant predictors. We found the ASA grade, and postoperative ICU admission as predictors of postoperative frailty. Compared to previous predictive models, this study is the first to include ASA grade, and postoperative ICU admission as predictors for postoperative frailty. The ASA grading system, developed by the American Society of Anesthesiologists (ASA), evaluates a patient’s overall health condition and ability to tolerate surgery preoperatively [32]. ASA grade and frailty are both significant factors influencing postoperative complications [33, 34]. Guidelines and previous research recommend combining the ASA grade with preoperative frailty assessments to predict patients’ postoperative outcomes and prognosis [33, 35, 36]. Although frailty is often observed in patients with higher ASA grades [37], there has been less discussion about the impact of ASA grade on postoperative frailty. This is partly due to the insufficient attention given to postoperative frailty in current clinical research. Additionally, previous studies have often focused on single disease types with similar patient characteristics, which may not show significant differences in terms of frailty development. However, our study results indicate that as the ASA grade increases, the likelihood of postoperative frailty also rises. A higher ASA grade is typically associated with more complex surgeries and poorer overall health, which makes individuals more prone to physical frailty.
ICU admissions is a important factor to frailty. The results of this study indicate that patients who were admitted to the ICU within seven days postoperatively have a higher likelihood of developing postoperative frailty. Studies indicated that ICU patients often present with metabolic disorders, and deficiencies in the synthesis of various hormones are associated with the onset of frailty in the older person [38, 39]. Additionally, factors such as mechanical ventilation and inflammation can lead to rapid muscle loss over a short period, thereby promoting the development of frailty [40]. Furthermore, the older person who are physically aging and admitted to the ICU are more susceptible to the lasting effects of complications, making them more prone to developing frailty [41]. This is consistent with the findings of Brummel, where 22% of patients with a history of ICU admission developed new-onset frailty within three months, and those who were already frail transited to a worse frailty state [42]. The findings of this study suggest that particular attention should be given to the prevention of postoperative frailty in older person who are at risk of ICU admission after surgery. Compared to previous studies, our model is applicable to a broad spectrum of surgical populations and is not limited to a single disease factor. It holds reference value for all older individuals who have undergone surgery.
In terms of physiological indicators, hemoglobin (Hb) concentration has been identified as a significant predictor of postoperative frailty. Hemoglobin levels reflect a patient’s anemic condition, and more severe anemia is generally associated with a higher incidence of frailty [43]. Anemia can reduce a patient’s oxygen-carrying capacity, leading to hypoxia in various tissues and organs, which in turn can affect daily activities and cognitive functions, thereby promoting the onset of frailty [44, 45]. However, it is important to note that the impact of anemia on frailty is most pronounced in moderate to severe cases, while mild anemia may not have as significant an effect on physical or psychological health. Patients with more severe anemia tend to be in poorer physical condition, which affects their daily activities, self-confidence, and adds to their psychological burden, contributing to the development of frailty. This aligns with findings from a cross-sectional study conducted in Asian community-dwelling older adults, which showed that for every 1 g/dL increase in hemoglobin, the odds of frailty were decreased by 6% after adjusting for covariates (OR = 0.94, 95% CI: 0.90–0.99) [46]. This suggests that clinicians should closely monitor hemoglobin levels, especially in patients with moderate to severe anemia, to identify those at higher risk of developing postoperative frailty.
Cognitive function is also an important component of postoperative frailty prediction. Our result is similar to previous studies, indicating that the reduction in cognitive function as a significant predictor of frailty [47]. This may be due to the similar pathogenic mechanisms shared by the two, both being influenced by chronic inflammation and oxidative stress [48]. Patients with impaired cognitive function experience a decline in their self-management abilities, which not only leads to reduced compliance but also exacerbates the progression of the disease, thereby increasing the risk of frailty. Our predictive model showed that low ADL scores and sedentary behavior were also associated with frailty. ADL and sedentary behavior are recognized as contributing factors to frailty in previous studies [49, 50]. Both ADL and sedentary behavior impact the muscular health of the older person. A decrease in activity can lead to a reduction in normal physiological functions, which in turn cause sarcopenia and osteoporosis, increasing the risk of frailty [50,51,52]. Additionally, a decline in mobility is associated with a range of chronic diseases, which can lead to the onset or exacerbation of existing conditions, causing a deterioration in physical health and increasing the likelihood of postoperative frailty [53, 54]. Therefore, it is crucial to assess perioperative cognitive and mobility capabilities to identify individuals at high risk of frailty and to intervene early.
The study reveals that social support factors, such as income and availability of caregivers, significantly predict postoperative frailty. Strong social networks and financial stability can improve patients’ quality of life and psychological health, potentially delaying frailty [55, 56]. In contrast, older person without support may undergo hospitalization and surgery with insufficient backing, affecting their mental health and ability to cope with illness [57]. Financial constraints can also deter older individuals from seeking necessary medical care, worsening their conditions and leading to frailty [56, 58]. Thus, special attention should be given to older person with limited support and financial resources, emphasizing the need for psychological support and daily care to mitigate postoperative frailty risks.
Current research on postoperative frailty is relatively scarce, and the development of predictive models for this condition is also limited. Li [10] and Liu [11] developed predictive models for postoperative frailty in elderly women (n = 403) undergoing percutaneous coronary intervention and elderly patients with fractures (n = 192). However, due to the limitations of a single-center sample size, disease specificity, and gender constraints, the clinical application value is limited. Our study increased sample size (n = 2290) and sample diversity through multicenter data collection, allowing our model to extend beyond postoperative predictions for a single disease or gender, focusing on a broader range of elderly surgical populations and enhancing the model’s versatility. The ample sample size also enabled us to divide into independent training and validation sets, further enhancing the model’s credibility through internal validation.
Our findings have important clinical implications and strengths. Firstly, frailty tends to progress and worsen, hence timely identification of high-risk groups is of significant importance [59]. Our model offers a novel approach to clinical frailty prediction, assisting healthcare professionals in the timely identification of patients at risk for postoperative frailty, enabling early intervention. Secondly, the dataset used in this study was obtained from seven Chinese institutions. The extensive sample size and multicenter origin of this dataset ensure its representativeness and generalizability in relation to the elderly surgical population in China. Lastly, our research fills a gap in the current literature regarding postoperative frailty, providing new insights into the occurrence and development of postoperative frailty. It’s worth noting that despite performing 1000 random data splits, the validation set still had a higher number of married individuals, higher PHQ scores, and shorter preoperative fasting times compared to the training set. However, these variables were not included in the final model. To ensure the objectivity of the results, we used 10-fold cross-validation during model testing. The ROC curve results showed that the model performed well in both the training and validation sets, so we consider these differences to be negligible.
Although our model performed well in bootstrap resampling and internal validation, external validation is still necessary to confirm its broad applicability. As an observational study, our results are subject to inherent biases. Future large-scale observational studies could further confirm the impact of risk factors on frailty, or randomized controlled trials (RCTs) could evaluate the intervention effects for high-risk individuals. Our study was based on the Chinese population, and the predictive performance may be influenced by factors such as racial differences, healthcare practices, and cultural background when applied to other countries. Although the study included a diverse surgical cohort, there were fewer cardiac surgery patients, and participants over 85 years of age were not included, which may limit the model’s applicability to older adults or cardiac patients. Given the different frailty patterns in individuals over 85, caution should be exercised when applying the model to this group. Future research will focus on detailed assessments of postoperative frailty trajectories and comparative analysis of changes in predictive factors.
We constructed and validated a predictive model for the frailty status of older person on the thirtieth postoperative day based on data from multiple centers. Our model encompasses various aspects, including surgical factors, lifestyle habits, and biochemical indicators, and demonstrates good predictive performance and calibration ability. The resulting nomogram can be used for the prevention of postoperative frailty in the older person, to timely identify high-risk groups for frailty, to delay or prevent the onset of frailty, thereby improving the quality of life for the elderly after surgery and promoting postoperative recovery.
During the current study period, the datasets that were utilized and/or analyzed may be obtained from the corresponding author upon reasonable request.
We express our gratitude for the cooperation of the seven hospitals involved in data collection and all the individuals who participated in the study.
The study was supported by the Institute of Science and Technology of the National Health Commission of the People's Republic of China (No.2021KYSHX016010201).
Ethical approval was obtained from the Institutional Review Board of the Second Affiliated Hospital of Guangzhou University of Chinese Medicine (protocol number BE2022-165-01). All participants signed a paper version of the informed consent form.
Not applicable.
The authors declare no competing interests.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
Lu, S., Liang, H., Fang, J. et al. Construction and validation of a risk prediction model for postoperative frailty in older adults:a multicenter study. BMC Geriatr 25, 88 (2025). https://doi.org/10.1186/s12877-025-05741-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12877-025-05741-y